|
This stage involves basic laboratory research and often lasts 2-4 years. First Steps: Laboratory and Animal Studies Regulation and oversight increase as the candidate vaccine makes its way through the process. The first stages are exploratory in nature. In the United States, vaccine development and testing follow a standard set of steps. Stages of Vaccine Development and Testing Many countries have adopted the WHO standards. A committee of the World Health Organization makes recommendations for biological products used internationally. In the European Union, the European Medicines Agency supervises regulation of vaccines and other drugs. It is now known as the Center for Biologics Evaluation and Research. Later, the DBS was renamed the Bureau of Biologics, and became part of the Food and Drug Administration. After a poliovirus vaccine accident in 1954 (known as the Cutter incident), the Division of Biologics Standards was formed to oversee vaccine safety and regulation. The United States Public Service Act of 1944 mandated that the federal government issue licenses for biological products, including vaccines. The Act established the government’s right to control the establishments where vaccines were made. The Hygienic Laboratory eventually became the National Institutes of Health. Public Health Service to oversee the manufacture of biological drugs. The Act created the Hygienic Laboratory of the U.S. This act emerged in part as a response to, which involved smallpox vaccine and diphtheria antitoxin. This was the first modern federal legislation to control the quality of drugs. Congress passed "An act to regulate the sale of viruses, serums, toxins, and analogous products," later referred to as the (even though "biologics" appears nowhere in the law). However, no regulation of vaccine production existed. They were smallpox, rabies, plague, cholera, and typhoid vaccines. Government Oversight In the United StatesĪt the end of the 19th century, several vaccines for humans were developed. 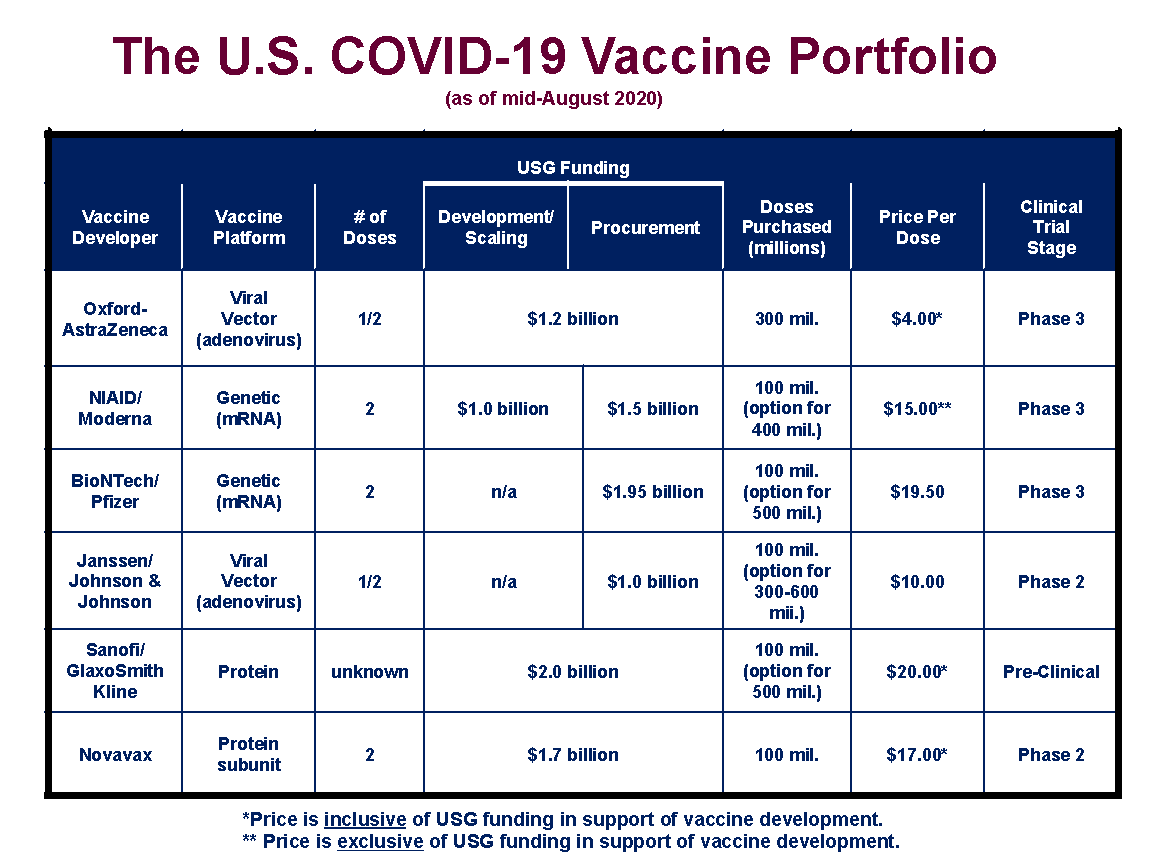
The current system for developing, testing, and regulating vaccines developed during the 20th century, as the groups involved standardized their procedures and regulations. Vaccine development is a long, complex process, often lasting 10-15 years, and involves a combination of public and private involvement. Please see the section on the COVID-19 pandemic and Operation Warp Speed for information on the COVID-19 vaccines. The article below mostly covers vaccines before the current COVID-19 pandemic. Pfizer and BioNTech are developing and testing new versions of their vaccine against recent variants, and might eventually alter their genetic recipe to mass-produce Covid-19 vaccines that target specific variants.EDITOR'S NOTE ( ): Many anti-vaccine people and organizations are not presenting the information in this article in full context. Many of the coronavirus variants now in circulation have key mutations in their spike proteins that help the virus bind more tightly to human cells or evade some kinds of antibodies. A facility in Baltimore run by Emergent BioSolutions had to throw out up to 15 million doses of Johnson & Johnson’s vaccine because of possible contamination. The single-dose Johnson & Johnson vaccine uses an adenovirus to carry DNA into human cells. (Moderna declined to provide filming access to their facilities.) 5, health care workers gave thousands of shots of the Moderna vaccine, which also uses mRNA to build immunity. The City of Los Angeles hosts a mass vaccination site at Dodger Stadium, above. 
More than a billion doses have been administered worldwide. 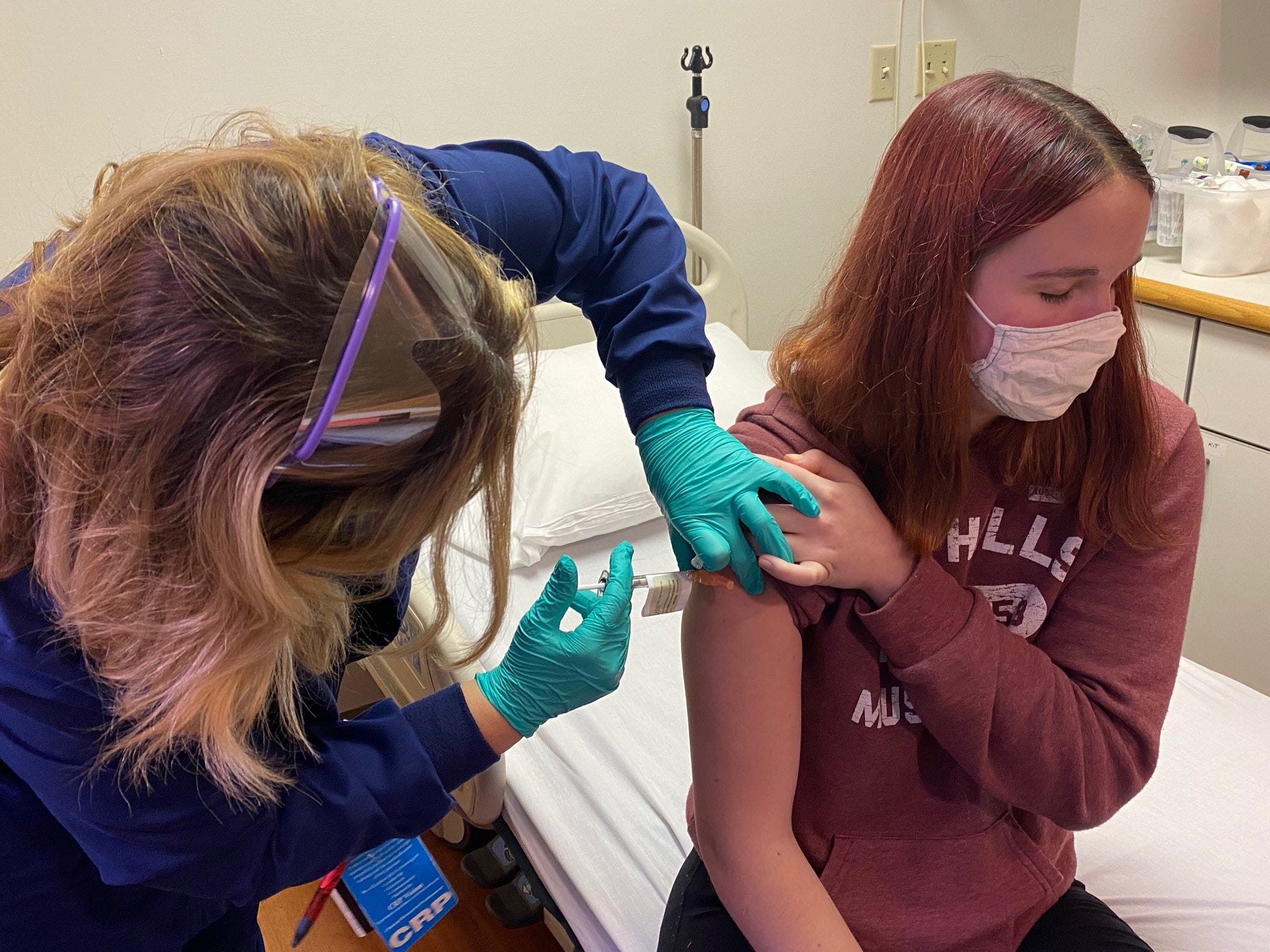
Some 141 million people in the United States - more than half of the nation’s adults - have received at least one dose of a Covid-19 vaccine. I gotta go help.” – Jacquelyn Chartier nurse vaccinator “I saw on the news that they were going to roll out these mass vaccination sites, but they needed nurses because they were short.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
